This Top Tip comes from Patricia Hugman, Senior Laboratory Technician at St Joseph’s College Toowoomba. Thank you, Patricia! This is something we get asked all the time and the collective wisdom of lab techs is the best source of info.
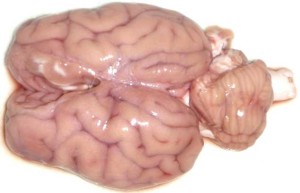
Have you ever wondered how long it would take to defrost … brains for dissection?
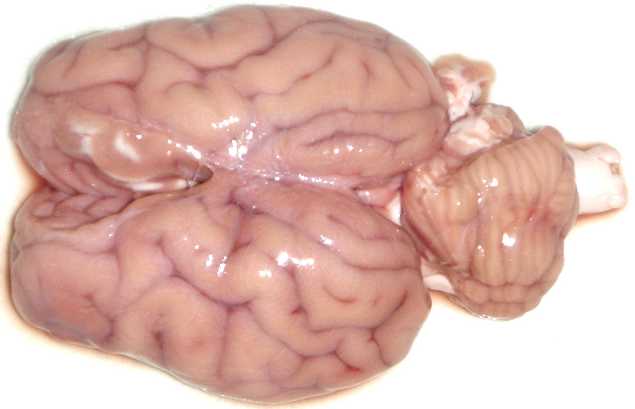
I found the following method for defrosting brains to be very effective & efficient
To defrost brains ‘al dente’ in 1hr
1. Straight from freezer
2. Place brains in packet in suitable container & cover with water (4 litre icecream container works well) sitting ‘flat’ not ‘end on’
1pkt/ container defrosts faster that 2 in one container
3. @20min – Change water
4. @40min – discard water, remove brains from packet, sprinkle liberally with salt, just cover with water
5. @50min – gently separate brains, being careful of brainstem/medulla
6. @60min – carefully remove individual brains, pat dry with paper towel and ‘serve’ to students!
On a nice summer’s day (25deg C) this timing worked a treat. Producing brains that were still firm but not icy.
Sitting on the bench (in air) produced irregular defrosting >4hr
Got a Top Tip you are willing to share with other techs? Have a squeal to Miss Vivi and see your name in lights.